From Spodumene To Battery-Grade Lithium - The Complete Processing Journey
Turning spodumene into battery-grade lithium is far more than a single step, but it’s a complex chain of thermal and hydrometallurgical processes. This post breaks down the full journey from ore to high-purity lithium compounds.

In the modern quest for a sustainable future, lithium has earned the title of "white gold". It is driven by the explosive growth of the electric vehicle (EV) market, projected to expand at a compound annual growth rate of 13%. To meet this demand, the world relies on two primary sources: continental brines and hard-rock minerals. While brines currently lead in production volume, spodumene is the undisputed champion of the mineral world due to its high lithium content and faster processing speed.
In this post, I will explain the story of how an inert, grey-white rock buried deep in the Earth's crust is transformed into the high-purity chemical that powers our smartphones, electric cars, and the majority of devices with batteries.
The Starting Point: Understanding the Spodumene "Refractory" Problem
Spodumene (LiAlSi2O6) is a lithium aluminum silicate mineral found in pegmatite deposits. In its natural state, it exists as α-spodumene, a monoclinic pyroxene with a compact, dense crystal structure (3.27 g/cm3).
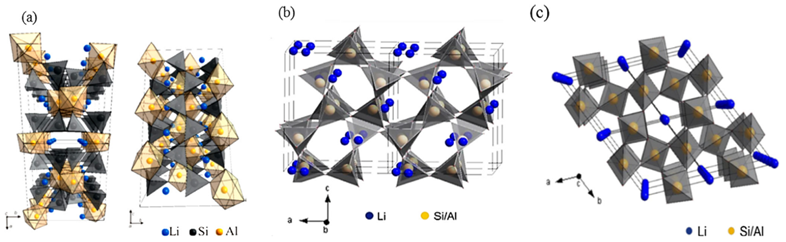
For a non-expert, think of α-spodumene as a tightly locked safe. The lithium atoms are trapped inside cavities between silicon and aluminum units, held by coulombic forces. This makes the mineral "refractory", meaning it is chemically inert and resistant to almost all acids and bases at room temperature. To get lithium out, you must first "unlock" the structure.
Phase 1: The Crystal Fortress - Mining and Beneficiation
The journey begins at the mine. Raw ore typically contains only 1% to 3% Li2O, mixed with "gangue" minerals like quartz, feldspar, and mica. The goal of beneficiation is to create a high-grade concentrate (usually 6% Li2O or approximately 75% spodumene).
- Crushing and Grinding: You crush the ore and mill it into fine particles to "liberate" the spodumene from surrounding waste rock.
- Heavy Media Separation (DMS): This process uses the difference in density between minerals; spodumene is denser than quartz and feldspar, allowing it to be separated in a "sink-float" tank.
- Froth Flotation: The most popular technique for fine particles. Chemicals are added to a tank of bubbles where the spodumene sticks to the bubbles and floats to the top, while the waste stays at the bottom.
- Magnetic Separation: This step removes iron-bearing minerals that could contaminate the final battery material.
Phase 2: The Great Transformation - Roasting and Decrepitation
Once you have 6% concentrate, you face the industry's biggest challenge: the α→β phase transformation.
The Traditional Way: Decrepitation at 1100 °C
For over 70 years, the standard method has been to roast the concentrate in a massive rotary kiln at 1000-1100 °C. At these extreme temperatures, the "safe" finally unlocks. The α-spodumene finally shatters and "unfolds" into β-spodumene.
This transformation is dramatic: the mineral undergoes a 30% volume expansion, becoming chalky, brittle, and highly reactive. However, this step is the "bottleneck" of the industry. It is incredibly energy-intensive and produces a massive carbon footprint, releasing approximately 9 tons of CO2 for every ton of lithium produced.

The "Green" way: Direct Extraction
Modern research is focused on bypassing the 1100 °C kiln entirely. Innovative methods now allow for the direct extraction of lithium from α-spodumene at much lower temperatures.
- NaOH Roasting (325 °C): A groundbreaking method roasts α-spodumene with NaOH at just 325 °C. This converts the mineral into water-soluble salts like LiNaSiO4.
- Solid-State Carbonate Reaction (750 °C): This is also called Direct Lithium Extraction (DLE) from α-spodumene with Na2CO3 and Al2O3 at 750 °C. In this innovative process, α-spodumene concentrate is mixed with Na2CO3 and Al2O3 in a specific 2:2:1 molar ratio. Instead of 1100 °C, the mixture is heated to only 750 °C for about 4 hours. The addition of Al2O3 is the secret ingredient as it acts as a "trap" for the excess silica that usually prevents lithium recovery, providing a four-fold increase in the thermodynamic driving force. This solid-state reaction converts the Li directly into Li2CO3 with a yield of over 90%, completely bypassing the energy-heavy phase-transformation step and the need for corrosive acids.
- Fluorine Chemical Method: Using hydrofluoric acid (HF) or fluoride salts can destroy the silicate bonds at even more moderate temperatures (100-157 °C), though safety concerns regarding toxicity remain high. Dealing with HF is no joke.
Phase 3: The Chemical Attack - Digestion and Leaching
After roasting, the lithium is no longer trapped in a rock. It is in a reactive chemical form. In the next stage, leaching, you dissolve the lithium into a liquid "pregnant leach solution" (PLS).
- Acid Baking (Traditional): The β-spodumene is mixed with concentrated sulfuric acid (H2SO4) at 250 °C. The acid's hydrogen ions kick out the lithium ions, creating water-soluble lithium sulfate (Li2SO4).
- Water Leaching: The resulting "bake" is mixed with water. The Li2SO4 dissolves into the water, while the unwanted aluminum and silicon stay behind as a solid residue.
- Alkaline Digestion: In newer processes, the concentrate is treated with lime (CaO) and NaOH under high pressure in an autoclave. This yields lithium hydroxide (LiOH) directly.
Phase 4: Purification - Removing the "Imposters"
The liquid PLS contains Li, but it also contains unwanted "impostors" (impurities) like iron (Fe), aluminum (Al), calcium (Ca), and magnesium (Mg). If even a tiny amount of these remains, the battery might fail or catch fire.
- Chemical Neutralization: First, the solution's pH is adjusted (often between 10-13, depending on the technology used) using lime or caustic soda. This causes Fe and Al to precipitate as hydroxides (turn back into solids) so they can be filtered out.
- The Magnesium Hurdle: Magnesium is the most difficult impurity because its physical properties are very similar to lithium. Modern plants often use nanofiltration (NF). Those advanced membranes act as molecular sieves, with nanometer-sized pores that allow small monovalent ions like lithium (Li+) to pass through while blocking larger, multi-charged ions like Mg2+ and Ca2+.
- Electrodialysis (ED): For ultra-high purity, techniques like electrodialysis use an electric field to pull Li+ ions through selective ion-exchange membranes, effectively concentrating the lithium while leaving residual salts behind.
- Polishing: Na2CO3 can also be used to precipitate calcium and magnesium. Often, ion exchange resins (high-tech chemical filters) are used for a final deep clean to ensure the solution is pure.
Phase 5: Precipitation - Creating the Final Product
Depending on the method used, you now have a nearly pure or crystal-clear, high-purity lithium solution. The final step is to precipitate the lithium as a solid salt that battery manufacturers can use.
- Producing Lithium Carbonate (Li2CO3): Soda ash (Na2CO3) is added to the purified lithium sulfate solution at 90-100 °C. Because lithium carbonate is less soluble in hot water, it "snows" out of the solution as white powder. White powder of lithium carbonate can contain small amounts of soda ash in it, which can be removed by washing with water.
- The Bicarbonation "Upgrade": To reach battery grade (>99.5%) purity, the carbonate is dissolved again using CO2 to form lithium bicarbonate (LiHCO3). When this solution is heated, only the purest lithium carbonate precipitates back out.
- Producing Lithium Hydroxide (LiOH): Some batteries require lithium hydroxide. This is produced by reacting lithium sulfate with sodium hydroxide and then cooling the solution to freeze out impurities like Glauber's salt (Na2SO4∙10H2O).
Phase 6: The "Zero-Waste" Goal: Valorizing the Residues
A major drawback of traditional processing is the huge amount of solid residue produced as waste. In a circular economy, you want to turn this waste into wealth in the form of a meaningful, commercialized byproduct with significant value.
Aluminum silicate residue usually contains mainly nepheline (NaAlSiO4) or analcime (NaAlSi2O6∙H2O). These materials are not actually waste, but highly valued in the glass and ceramics industries as substitutes for feldspar. They are even used in manufacturing dental porcelain, tableware, and high-performance glazes.
Conclusion
The future, from a hard-rock pegmatite to a high-purity battery chemical, is an engineering marvel. While the industry has long relied on the high-temperature "decrepitation" method, the future is moving towards direct alkali roasting and hydrothermal conversion. These technologies not only reduce energy costs but also pave the way for a truly sustainable lithium supply chain; one that minimizes waste, reduces CO2 emissions, and ensures that the "white gold" of the 21st century is as green as the cars it powers.
Which step in the lithium processing chain do you think is the most challenging to scale, and why?
Follow for more insights on lithium, hydrometallurgy, and critical raw materials.