What Is Hydrometallurgy And Why Does It Matter For Battery Supply Chain?
Hydrometallurgy sits at the core of modern metal extraction, especially for battery materials. From leaching to purification, these processes define whether lithium and other critical metals can reach battery-grade quality.

In the race to decarbonize the global economy, lithium has earned the moniker "white gold". As nations commit to phasing out internal combustion engines, with the EU scheduling a ban on petrol and diesel cars for 2035, the demand for lithium-ion batteries (LIBs) is a massive leap from just 390 kilotons in 2020 to over 1.6 million tons by 2030, and the supply chain faces a massive challenge. To meet this demand sustainably, the world is turning to a sophisticated branch of extractive science: hydrometallurgy.
Understanding Hydrometallurgy: The Science of Aqueous Solutions
Hydrometallurgy is a method of obtaining metals from ores, concentrates, and recycled materials using aqueous (water-based) chemistry. Unlike traditional smelting, which uses extreme heat, hydrometallurgy operates through a sequence of chemical reactions in solution. The process typically follows two critical stages:
- Leaching: The first and most crucial step, where targeted metal ions (like Li, Co, Ni, and Mn) are dissolved into a liquid medium using reagents such as acids, bases, or even microorganisms. But water is the most favorable medium.
- Extraction and purification: Once the metals are in the "pregnant leach solution" (PLS), they must be isolated. This is achieved through techniques like solvent extraction, chemical precipitation, and ion exchange to produce high-purity metal salts suitable for battery production.
Why Hydrometallurgy is Winning the Sustainability Battle?
For decades, pyrometallurgy, a process involving high-temperature smelting (>1400 °C), was industry standard. However, hydrometallurgy is increasingly favored for the modern battery supply chain for several reasons:
- Higher purity: Hydrometallurgical routes are capable of producing battery-grade lithium carbonate and hydroxide (often >99.5% purity) with far fewer impurities than smelting.
- Energy Efficiency: While pyrometallurgy is energy-intensive and releases a significant amount of greenhouse gases, hydrometallurgy operates at much lower temperatures, significantly reducing the carbon footprint of the extraction process.
- Lithium Recovery: One of the greatest flaws of smelting is that lithium often becomes trapped in the solid "slag," where it is difficult or impossible to recover. Hydrometallurgy, by contrast, allows for near-total recovery of lithium alongside other valuable metals like cobalt and nickel.

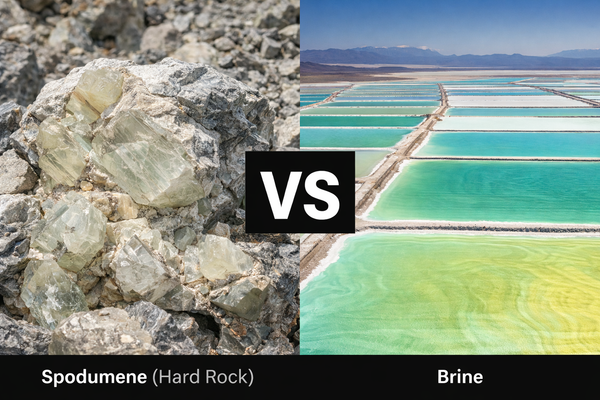
Breaking the "α-phase" Bottleneck in Spodumene
Most of Europe's and Australia's lithium is found in hard-rock spodumene. Historically, this has been harder to produce than South American brines because natural α-spodumene is chemically inert. Traditional processing requires "decrepitation," the ore at a staggering 1100 °C to convert it into a more reactive β-phase.
New hydrometallurgical innovations are bypassing this bottleneck. For example, a "groundbreaking" two-stage NaOH roasting method can achieve over 99% lithium recovery at only 325 °C, eliminating the need for the 1100 °C decrepitation and the subsequent hazardous sulfuric acid baking steps. Similarly, solid-state reactions using sodium carbonate and alumina can extract over 90% of lithium at 750 °C, relying only on a water wash to isolate the final product.
The Challenge of Separation: The Mg/Li Hurdle
One of the greatest technical challenges in the lithium supply chain is the separation of Mg from Li. They have very similar physical properties, which often lead to "co-precipitation", where Mg impurities ruin the quality of the Li product.
Hydrometallurgical tools like nanofiltration (NF) and electrodialysis (ED) are revolutionizing this. NF membranes act as molecular sieves that can differentiate between ions based on size and charge, effectively blocking magnesium while letting lithium pass through. This allows for the exploitation of "lower-grade" brines, such as those found in China's Qinghai-Tibet plateau, that were previously considered too difficult to process due to high Mg/Li ratios.
The "Green" Frontier: Organic Acids and Bioleaching
To further reduce environmental impact, researchers are exploring even "greener" lixiviants. While inorganic acids like sulfuric acid and hydrochloric acid are efficient, they can be corrosive and produce toxic byproducts like chlorine gas.
Organic acid leaching, using citric, oxalic, or metallic acids, offers a biodegradable and less corrosive alternative. For example, using citric acid has achieved over 95% leaching efficiency for Li, Ni, Co, and Mn. Even more radical is bioleaching (or biohydrometallurgy), which uses microorganisms like Aspergillus niger or Acidithiobacillus ferrooxidant to secret natural acids that dissolve metals. While currently slower than chemical methods, bioleaching is up to 50% more economical and represents a truly circular approach to mineral processing.
Securing the Chain: Recycling and the Circular Economy
Hydrometallurgy isn't just for mining. It is the backbone of the circular economy. As millions of EVs reach the end of their lives, we are entering an era of "urban mining" where spent batteries become a source of Li, Co, and Ni.
Recycling via hydrometallurgical routes can reduce greenhouse gas emissions by 17-61% compared to primary mining. Pyrometallurgical recycling often loses Li in the "slag" (waste byproduct) due to its high oxygen affinity. In contrast, hydrometallurgical recycling uses acid or organic leaching to dissolve the "black mass" of the battery, allowing for the recovery of lithium carbonate with efficiencies often exceeding 90%. The recycled lithium requires significantly less energy to produce than virgin lithium mined from the earth. By establishing a domestic "mine-to-battery" recycling loop, regions like Europe can reduce their strategic dependence on concentrated imports from China, Chile, and Australia.
Why It Matters for the Future
The transition to a sustainable battery supply chain is more complex than simply digging holes in the ground. It requires the precise, selective, and energy-efficient chemical engineering that only hydrometallurgy can provide. From the industrial-scale Metso-Outotec soda ash process (set in Finland) to low-temperature alkali roasting, these technologies are transforming lithium from a resource-heavy commodity into a sustainable cornerstone of the green energy era. The success of the battery supply chain depends on more than just finding lithium. It depends on purifying and recycling it efficiently.
Hydrometallurgy matters because it offers a pathway to:
- Faster production through direct lithium extraction (DLE)
- Higher sustainability by reducing water and energy usage
- Greater independence by allowing countries to recycle their own battery waste.
As we move toward a carbon-neutral world, hydrometallurgy will be the invisible engine driving the batteries that power our future.
Where do you see the biggest bottleneck in hydrometallurgical processing today: leaching, separation, or purification? Do you think it will continue to be the backbone of the circular economy?
Follow for more insights on lithium, hydrometallurgy, and critical raw materials.