Spodumene vs Brine - The Real Lithium Extraction Battle
Spodumene and brine follow fundamentally different paths to lithium — from energy use to processing complexity. Understanding these differences is key to evaluating the future of lithium supply.
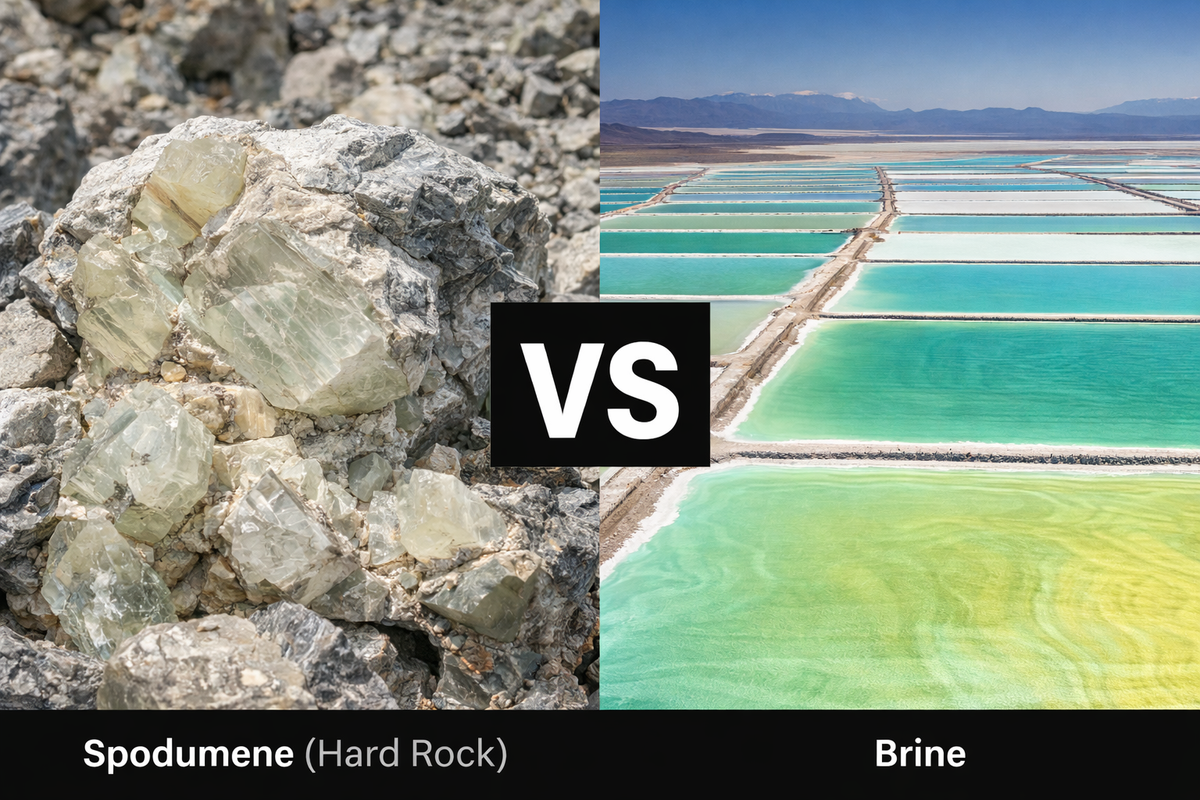
The global transition to clean energy has elevated lithium from a niche industrial metal to a critical strategic resource, sparking an intense extraction battle between brine, deposits, and hard-rock minerals like spodumene. Currently, continental brines account for approximately 60% of total global lithium production, while the remainder is largely derived from pegmatites, with spodumene being the most economically significant mineral source. The battle is defined by a complex trade-off between production costs, environmental sustainability, and speed to market.
The Brine Advantage: Low Cost and Scale
Lithium-enriched brines are primarily concentrated in the "Lithium Triangle" of Bolivia, Chile, and Argentina, which holds about 66% of the world's lithium resources. The extraction process typically relies on solar evaporation ponds, where sunlight concentrates the lithium over a period of 12 to 18 months. This method is favored for its low operational costs and relative simplicity, as it harnesses natural solar radiation rather than expensive chemical reagents for initial concentration.
However, brine extraction faces a significant hurdle, most notably the presence of magnesium (Mg) impurities, which are difficult to separate when the Mg/Li ratio is high. Furthermore, the long evaporation time makes brine production slow to respond to sudden spikes in market demand. Geographically, concentrated brine deposits are rare and highly regionalized, which creates geopolitical supply chain vulnerability for regions like Europe and Asia that lack large-scale salt flats.
The Hard-Rock Surge: Speed and Strategic Autonomy
In response to these geographical limits, interest has surged in hard-rock mining, particularly in Western Australia, which has become a world leader in spodumene production.
Spodumene (LiAlSi2O6) is a monoclinic pyroxene that typically contains between 1-3% Li2O in raw ore, and can be concentrated to over 6% for industrial processing. Unlike brines, spodumene can be produced into battery-grade lithium carbonate or hydroxide in a matter of days rather than years, offering much higher industrial agility.
Despite this speed, traditional hard rock processing is notoriously energy-intensive. Natural α-spodumene is chemically inert (mostly) and must undergo calcination (decrepitation) at 1000-1100 °C to transform into the more reactive phase, β-spodumene. This high-temperature step is the "bottleneck" of the process, contributing to a carbon footprint approximately three times larger than that of brines.
The Environmental and Waste Battleground
The real battle is increasingly fought on environmental grounds, where brines and spodumene present different ecological challenges. Brine extraction is highly water-consumptive in regions with rigid regions, requiring approximately 2 million liters of water per ton of lithium, with 95% of that lost to evaporation before precipitation even begins. This creates severe water scarcity and disrupts fragile local ecosystems.
Spodumene mining, however, is much more waste-intensive, generating eight times more solid waste than brine extraction. In terms of mass efficiency, about 6% of the extracted material is actually lithium, leaving the remaining 94% as tailings that often contain hazardous reagents like sulfuric acid. Hard rock mining also results in significant land disruption and potential contamination, whereas brine is a subsurface fluid.
Technological Innovations: Green Extraction and Direct Lithium Extraction (DLE)
To improve the sustainability of the hard-rock route, researchers are developing green extraction pathways that bypass the 1100 °C calcination. The two-stage NaOH roasting can achieve over 99% lithium recovery at only 325 °C, while solid-state reactions with Na2CO3 and Al2CO3 yield over 90% recovery at 750 °C without using toxic acids. Other hydrothermal methods can synthesize hydroxysodalite zeolite as a valuable industrial byproduct, potentially turning waste residues into profit.
On the brine side, the industry is moving toward Direct Lithium Extraction (DLE), which uses selective adsorbents, ion exchange resins, or membranes to capture lithium directly from the liquid. DLE technologies are rapid and efficient (up to 95% recovery) and completely avoid the need for massive evaporation ponds, significantly reducing the environmental footprint and land use.
Conclusion: A Diversified Future
Spodumene vs. brine is not a battle of one replacing the other, but rather a strategic diversification of the lithium supply chain. Brines will remain the cost leader for large-scale production, provided their water use and impurity challenges are addressed via DLE. Meanwhile, spodumene will continue to expand as the preferred source for strategic autonomy in Europe and Australia, especially as low-temperature, acid-free "green" extraction methods move toward industrial scale.
Ultimately, the winner of this battle will be the sector that most effectively balances high extraction efficiency with the rigid environmental standards of the emerging green economy.
What’s your view? Which route will dominate in the long term, especially knowing the geolocations of these sources across the globe?
Follow for more insights on lithium, hydrometallurgy, and critical raw materials.




